 Problem #8: At time zero, there are 10.0 grams of W-187. How much time will elapse for this sample to be considered gone? A radioactive sample is considered to be completely decayed after 10 half-lives. Problem #7: Fermium-253 has a half-life of 0.334 seconds. Problem #6: How long will it take for a 40.0 gram sample of I-131 (half-life = 8.040 days) to decay to 1/100 its original mass? N = 0.91254 (this is how many half-lives have elapsed) Solution: 17/32 = 0.53125 (this is the decimal amount that remains) Problem #5: A radioactive isotope decayed to 17/32 of its original mass after 60 minutes. Video: An Alternate Solution to the Above Problem What is the half-life of the sample?Ģ) How many half-lives must have elaspsed to get to 0.015625 remaining? Problem #4: After 24.0 days, 2.00 milligrams of an original 128.0 milligram sample remain. In addition, note that the question asked for the amount that decayed, not the amount that remaning. Note that the length of the half-life played no role in this calculation. How many grams of a 10.0 gram sample would have decayed after exactly three half-lives? 
Problem #3: Os-182 has a half-life of 21.5 hours. (1/2) 5.56 = 0.0213 (the decimal fraction remaining after 5.56 half-lives) If one had 6.02 x 10 23 atoms at the start, how many atoms would be present after 20.0 days? 
Problem #2: Pd-100 has a half-life of 3.6 days. (1/2) 3 = 0.125 (the amount remaining after 3 half-lives) If one had 100.0 g at the beginning, how many grams would be left after 7.2 minutes has elapsed? Problem #1: The half-life of Zn-71 is 2.4 minutes. III is false.ChemTeam: Half-Life Problems #1 - 10 Half-Life Problems #1 - 10 Ten Examples Problems involving carbon-14 Probs 11-25 Problems involving uranium-238 Probs 26-40 Examples and Problems only (no solutions) Return to Radioactivity menu Thus, decrease in masses are not equal in first and second decay. Since the mass after second decay is smaller than initial mass and rate of decay is directly proportional to mass, rate of decay after second decay is smaller than initial value. When mass decreases from m 0 to m 0/4, it does two half life Amount of matters decay in first half decay and second half decay are equal. 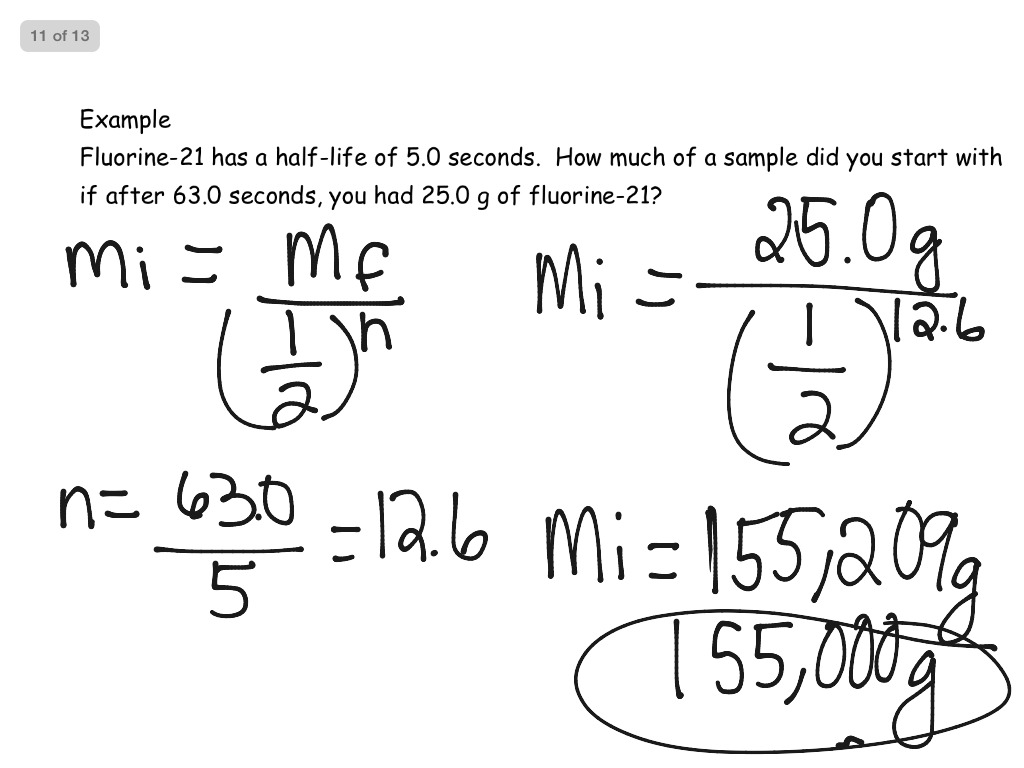
Rate decay of matter after 20 years is smaller than initial rate decay. Use this graph and state whether the following statements are true or false Where k is rate of decay and t (1/2) is half life.Įxample: Graph given below shows radioactive decay of matter X. Relation between half life and rate of decay is Since mass of matter decreases in decay process, rate of decay also decreases with time.Rate of decay is directly proportional to mass of radioactive matter.If masses of two matters are equal than matter having smaller half life has higher rate of decay. Rate of decay is inversely proportional to half life of matter.Rate of decay depends on half life and mass of matter. Rate of decay is number of disintegrated nucleus in unit time. It changes with the changes of types of matters.Īs we mention before, half life of matter is only depends on types of matter. It changes with changes in the temperature. It changes with the change in mass of matter. If initial mass is m 0 Īs you can see from the picture, there are 5 half life Įxample: Which ones of the following statements are true for half life of matters 
Since 31/32 of matter decays, 1/32 of matter is left.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
